cat no | io1013
ioGlutamatergic Neurons
PRKN R275W/WT
Human iPSC-derived Parkinson's disease model
ioGlutamatergic Neurons PRKN R275W/WT are opti‑ox deterministically programmed glutamatergic neurons carrying a genetically engineered heterozygous mutation in the PRKN gene encoding the Parkin protein. These cells offer a rapidly maturing, disease relevant and isogenic system for investigating the molecular and cellular significance of a heterozygous R275W mutation in Parkinson’s disease.
Related disease model cells are available now with a homozygous PRKN R275W mutation, and both can be used alongside their genetically matched control, ioGlutamatergic Neurons.
Place your order
Confidently investigate your phenotype of interest across multiple clones with our disease model clone panel. Detailed characterisation data (below) and bulk RNA sequencing data (upon request) help you select specific clones if required.
per vial
A maximum number of 20 vials applies. If you would like to order more than 20 vials, please contact us at orders@bit.bio.
Benchtop benefits
Make True Comparisons
Pair the ioDisease Model Cells with the genetically matched wild-type ioGlutamatergic Neurons to directly investigate the effect of heterozygous expression of mutant Parkin protein on disease.
Scalable
With opti-ox technology, we can make billions of consistently programmed cells, surpassing the demands of industrial workflows.
Quick
The disease model cells and isogenic control are experiment ready as early as 2 days post revival, and form structural neuronal networks at 11 days.
Highly characterised and defined
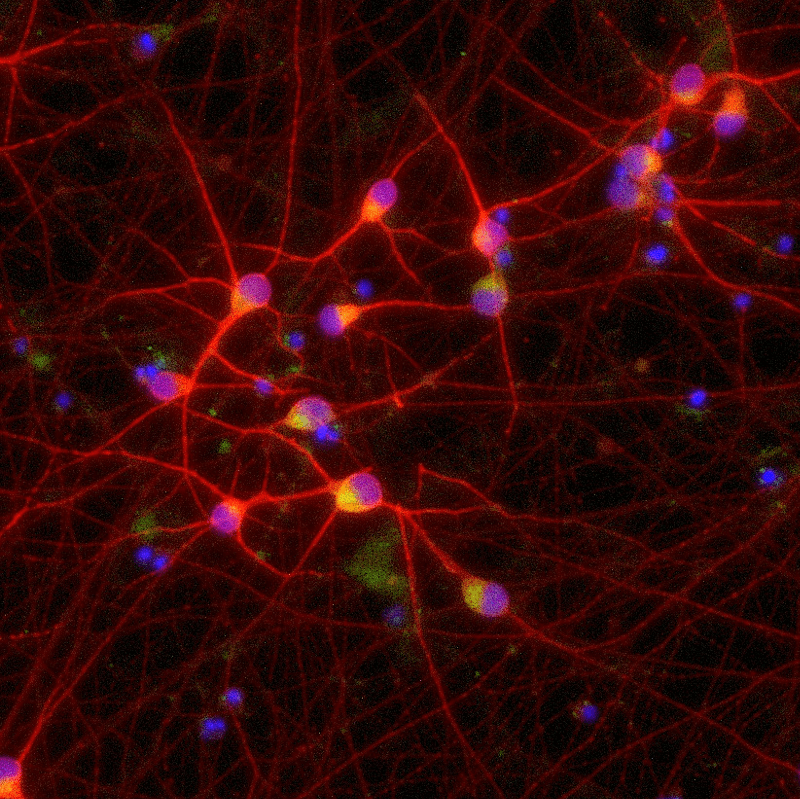
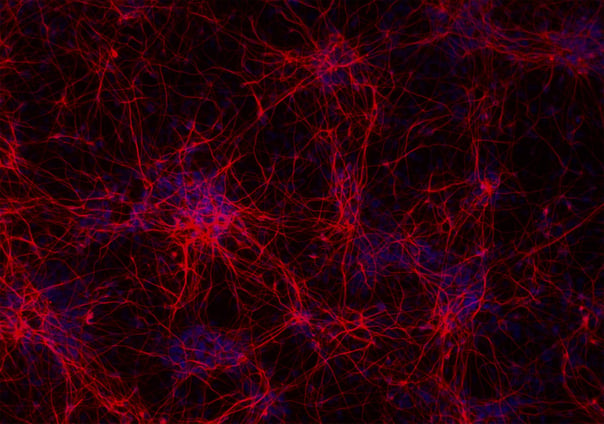
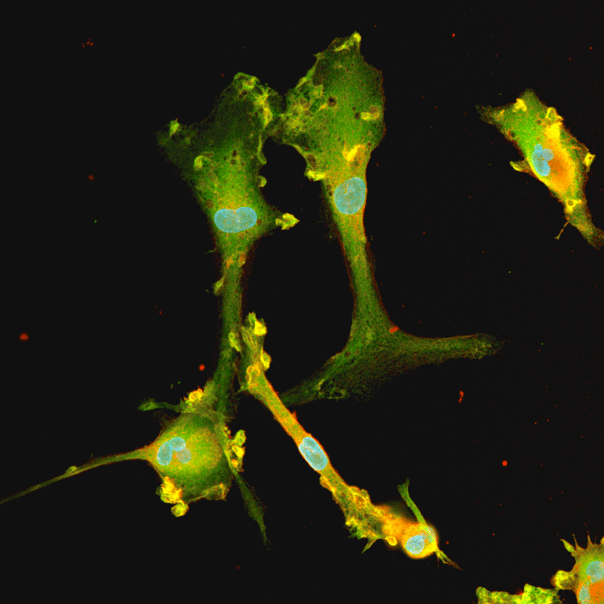
ioGlutamatergic Neurons PRKN R275W/WT express neuron-specific markers comparably to the isogenic control
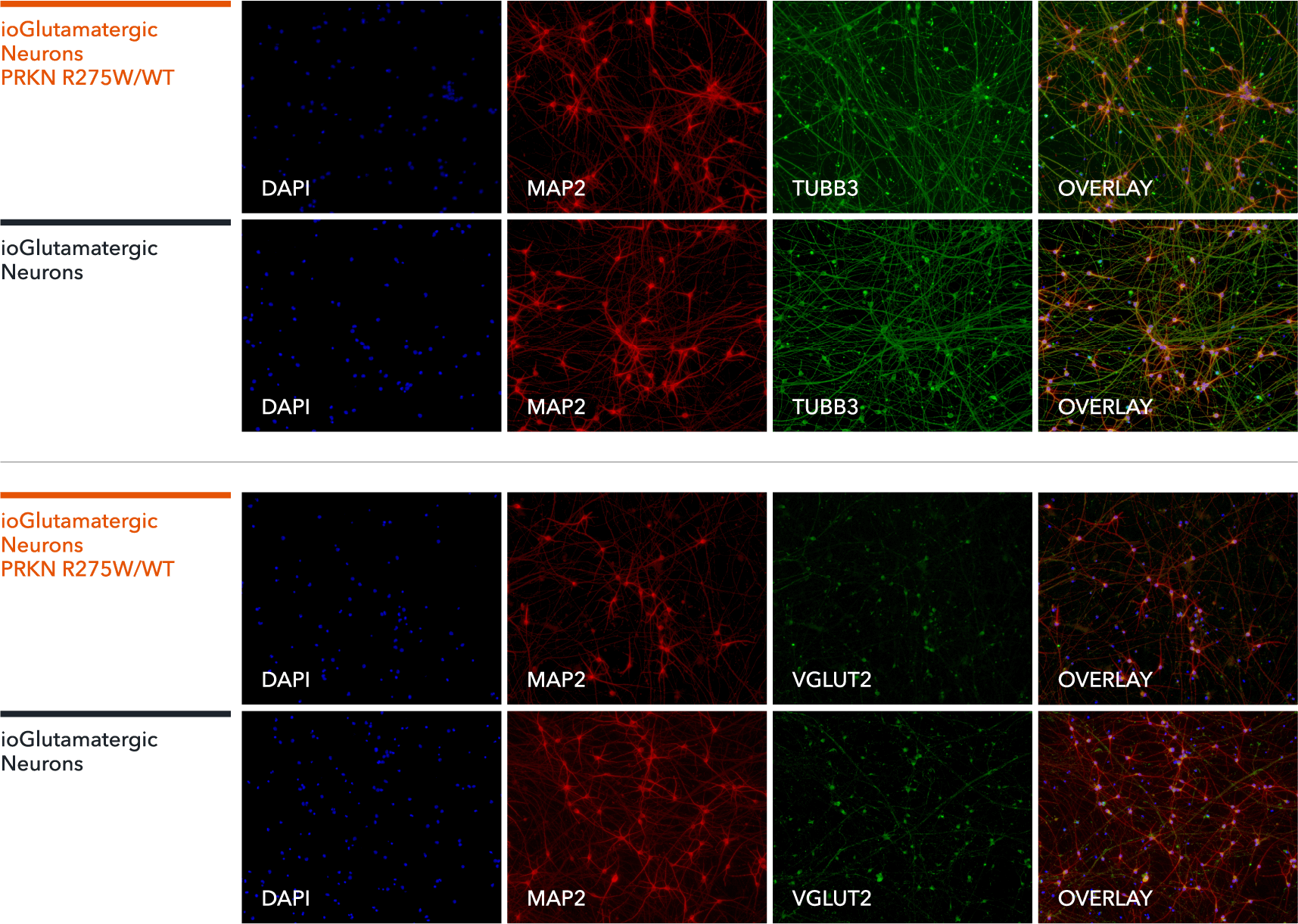
Immunofluorescent staining on post-revival day 11 demonstrates similar homogenous expression of pan-neuronal proteins TUBB3 and MAP2 (upper panel) and glutamatergic neuron-specific transporter VGLUT2 (lower panel) in ioGlutamatergic Neurons PRKN R275W/WT compared to the isogenic control. 100X magnification.
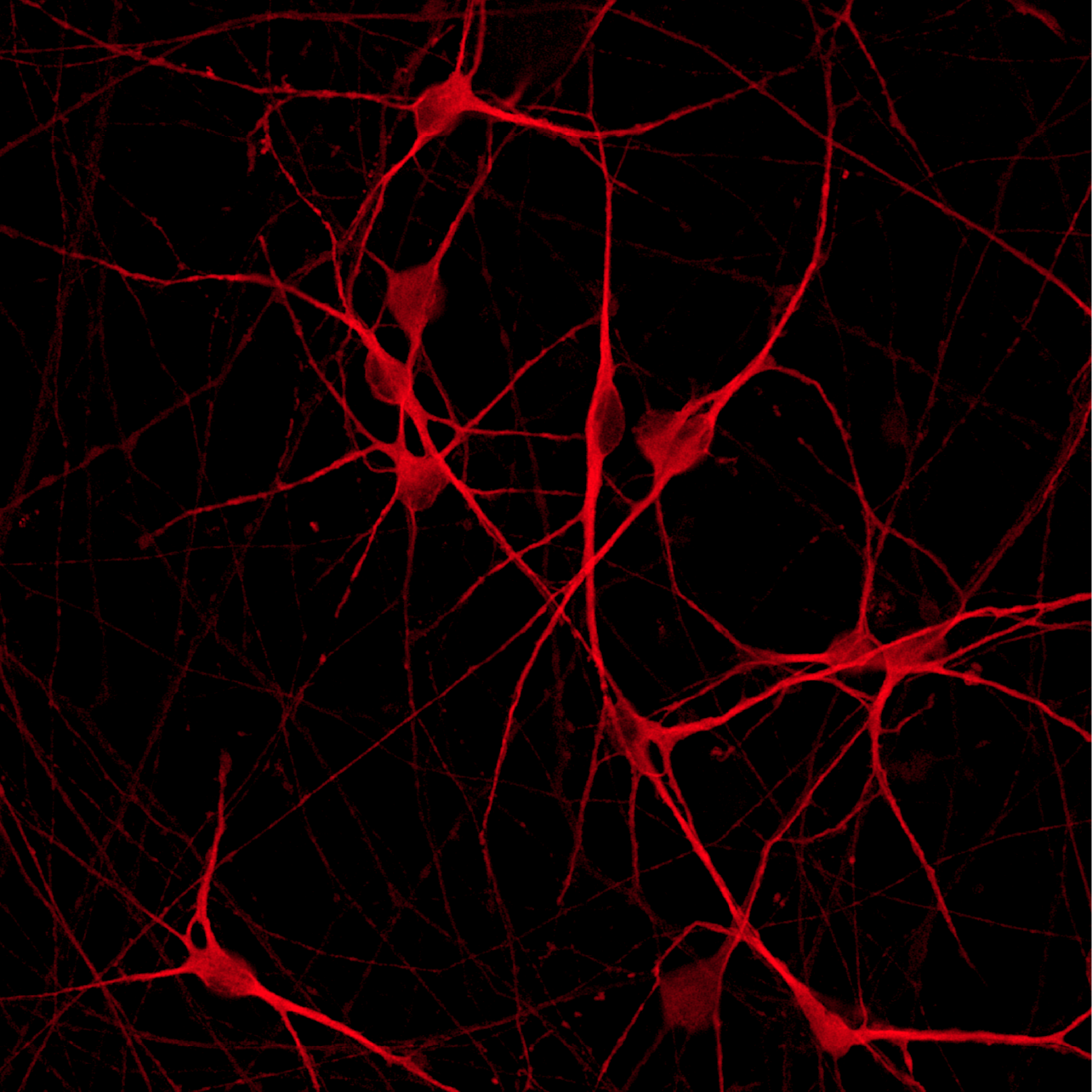
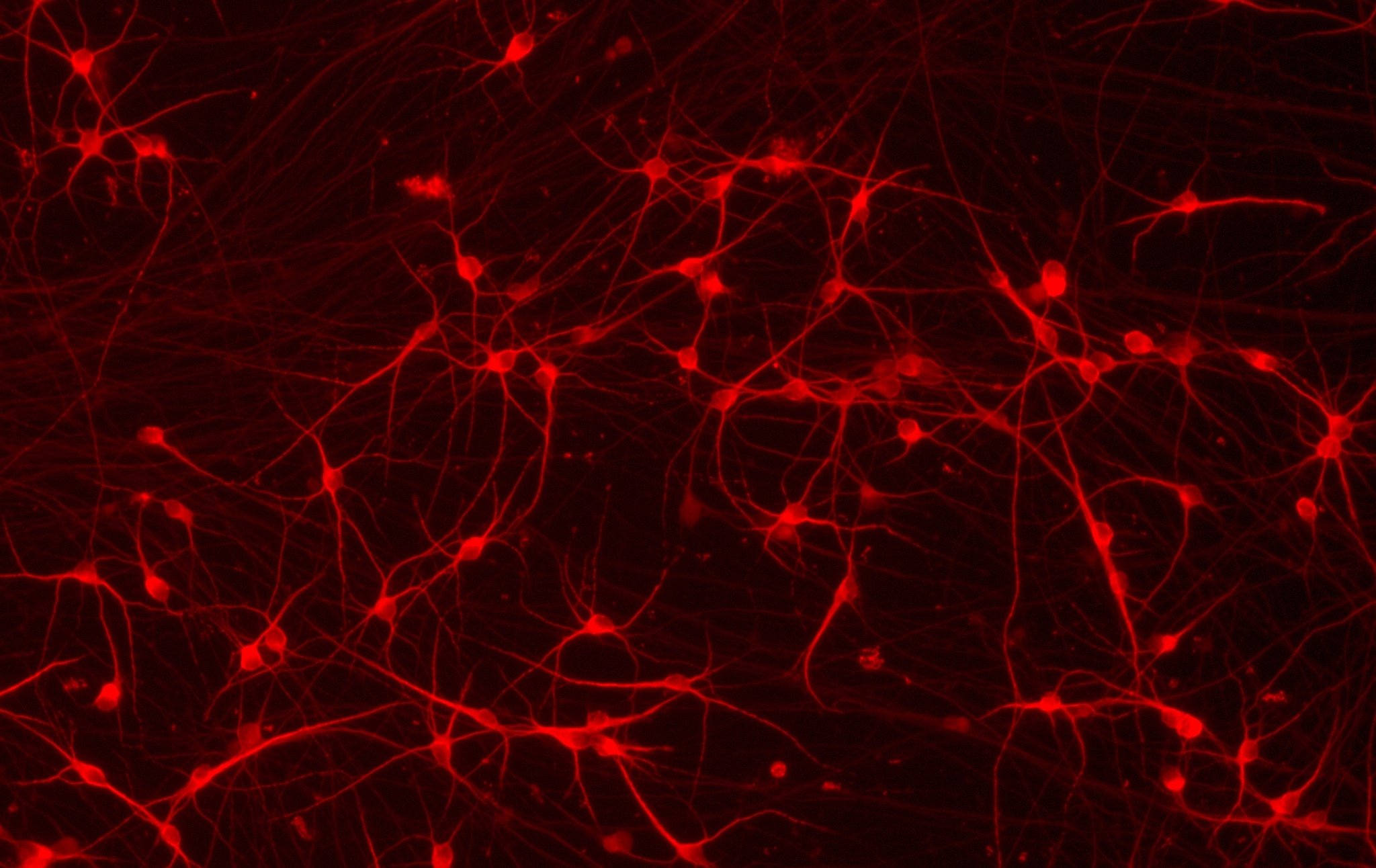
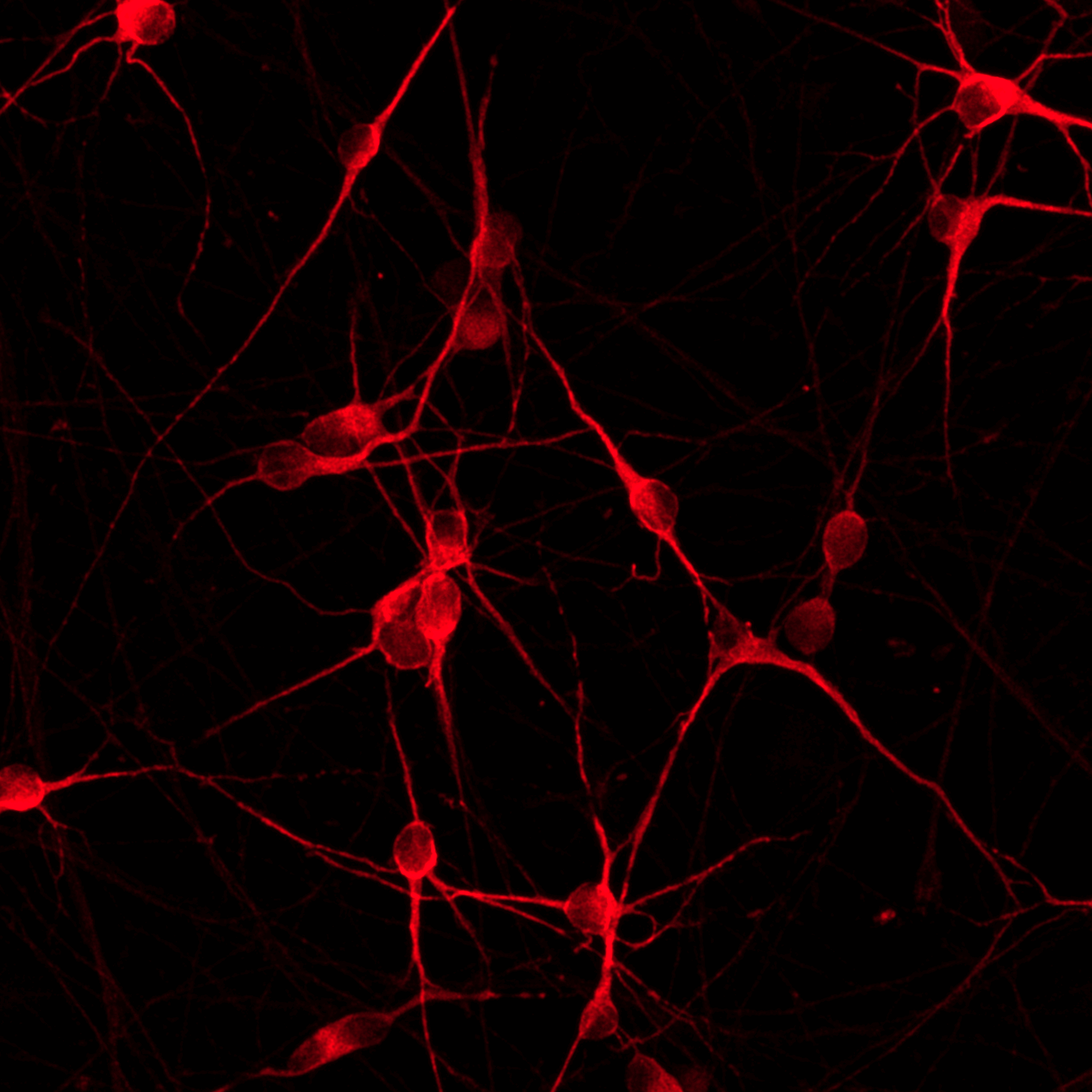
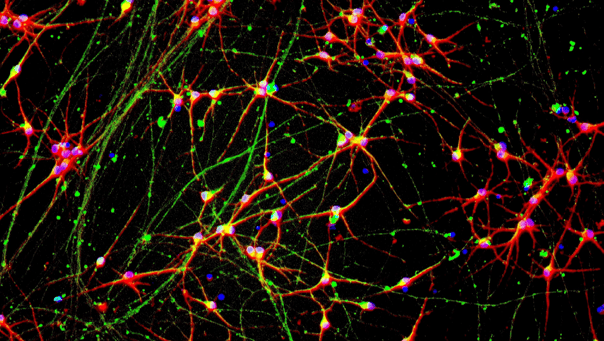
ioGlutamatergic Neurons PRKN R275W/WT form structural neuronal networks by day 11
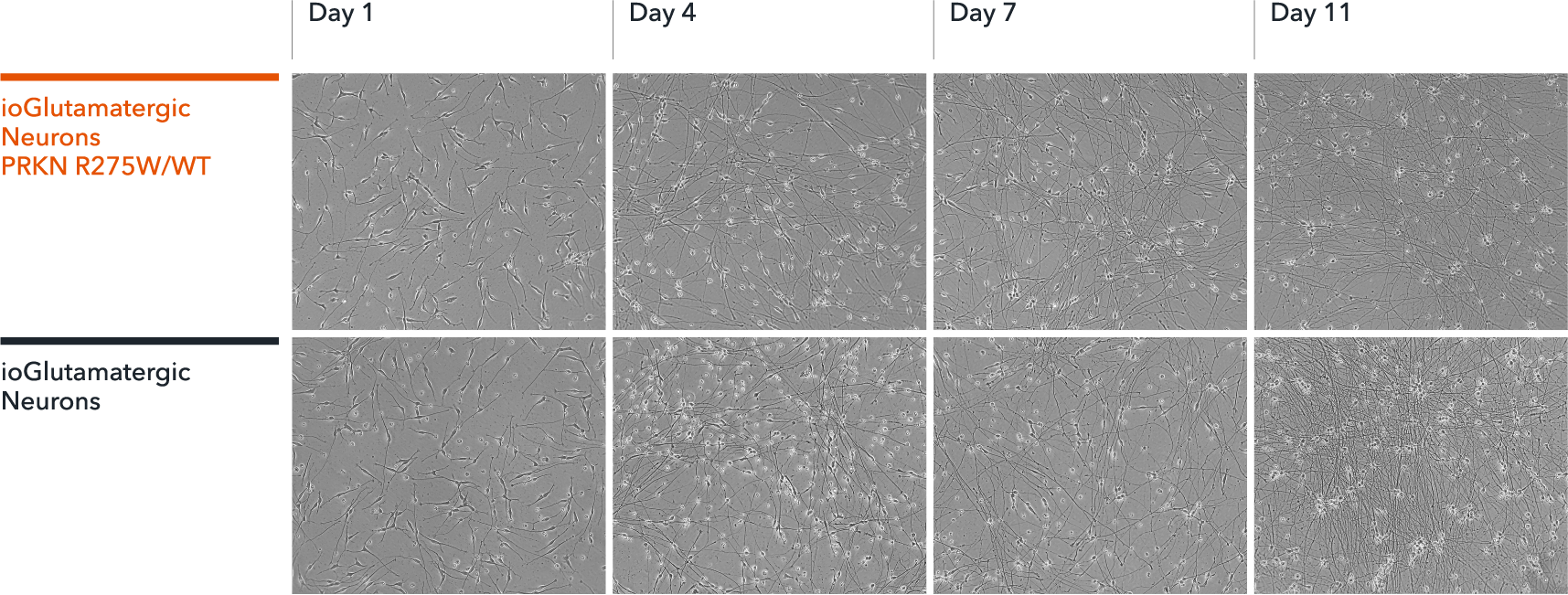
ioGlutamatergic Neurons PRKN R275W/WT mature rapidly and form structural neuronal networks over 11 days, when compared to the isogenic control. Day 1 to 11 post thawing; 100X magnification.
ioGlutamatergic Neurons PRKN R275W/WT demonstrate gene expression of neuronal and glutamatergic-specific markers following deterministic programming
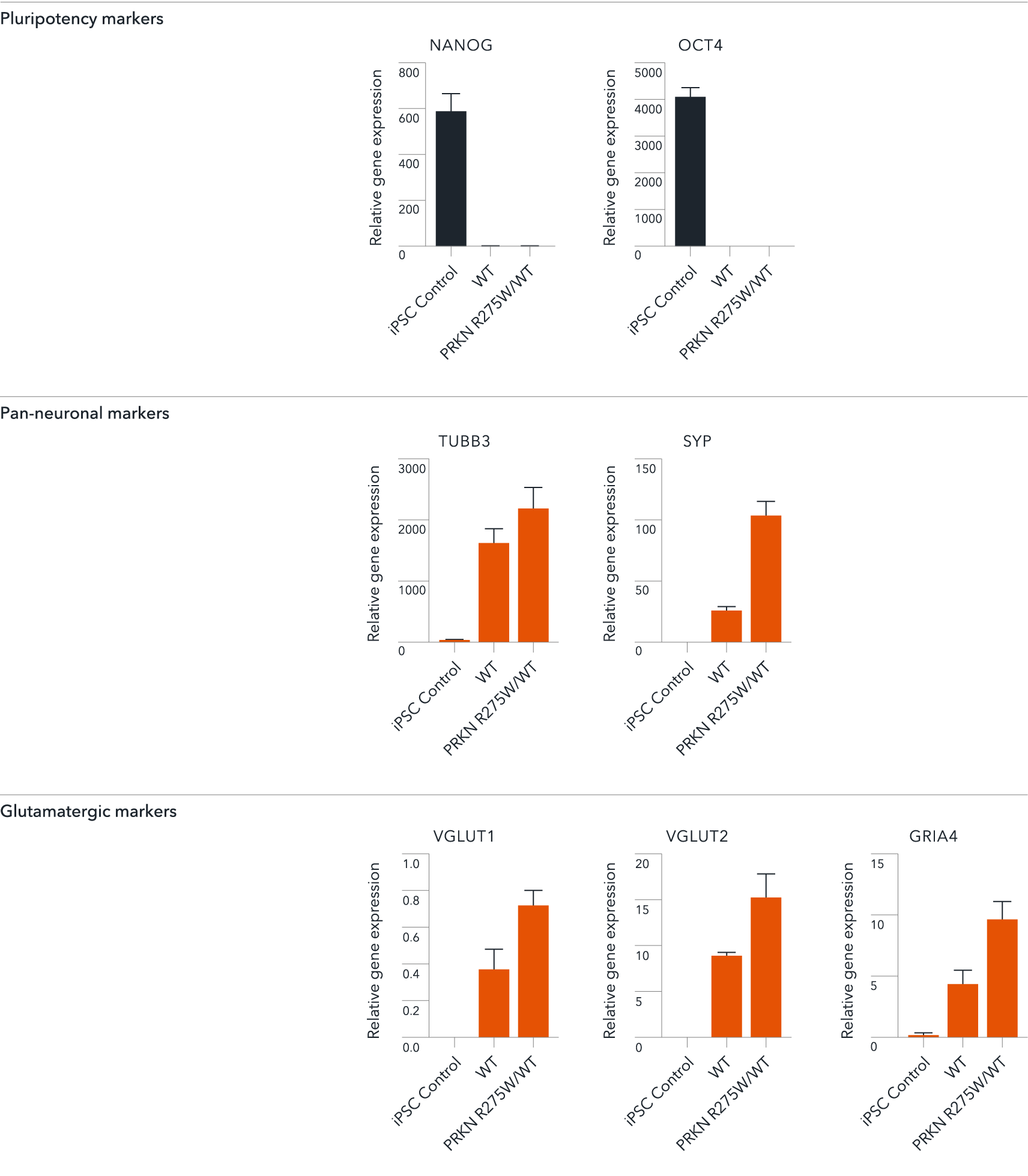
Gene expression analysis demonstrates that ioGlutamatergic Neurons PRKN R275W/WT and the isogenic control (WT) lack the expression of pluripotency markers (NANOG and OCT4) at day 11, whilst robustly expressing pan-neuronal (TUBB3 and SYP) and glutamatergic specific (VGLUT1 and VGLUT2) markers, as well as the glutamate receptor GRIA4. Gene expression levels were assessed by RT-qPCR (data expressed relative to the parental hiPSC control (iPSC Control), normalised to HMBS). Data represents day 11 post-revival samples.
Disease-related PRKN is expressed in ioGlutamatergic Neurons PRKN R275W/WT following deterministic programming
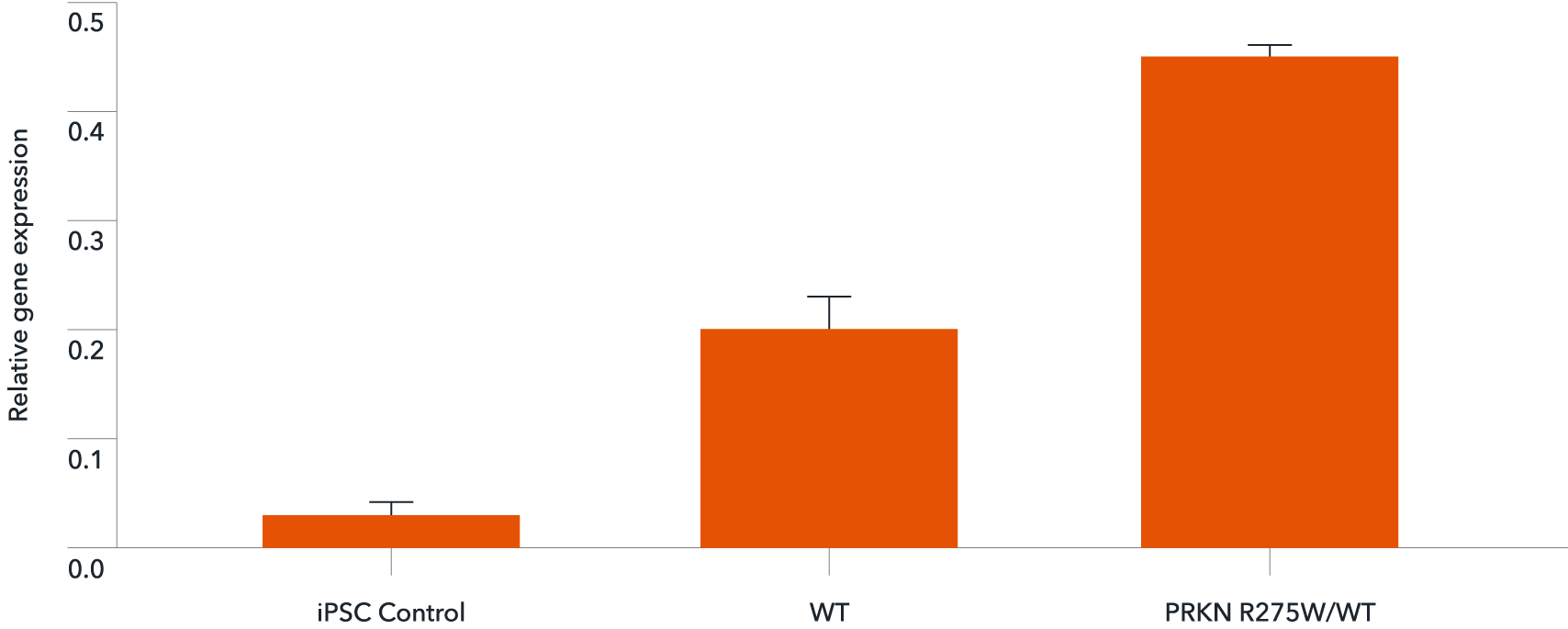
Gene expression analysis demonstrates that ioGlutamatergic Neurons PRKN R275W/WT and the isogenic control (WT) express the PRKN gene encoding the Parkin protein. Gene expression levels were assessed by RT-qPCR (data expressed relative to the parental hiPSC control (iPSC Control), normalised to HMBS). Data represents day 11 post-revival samples.
Cells arrive ready to plate
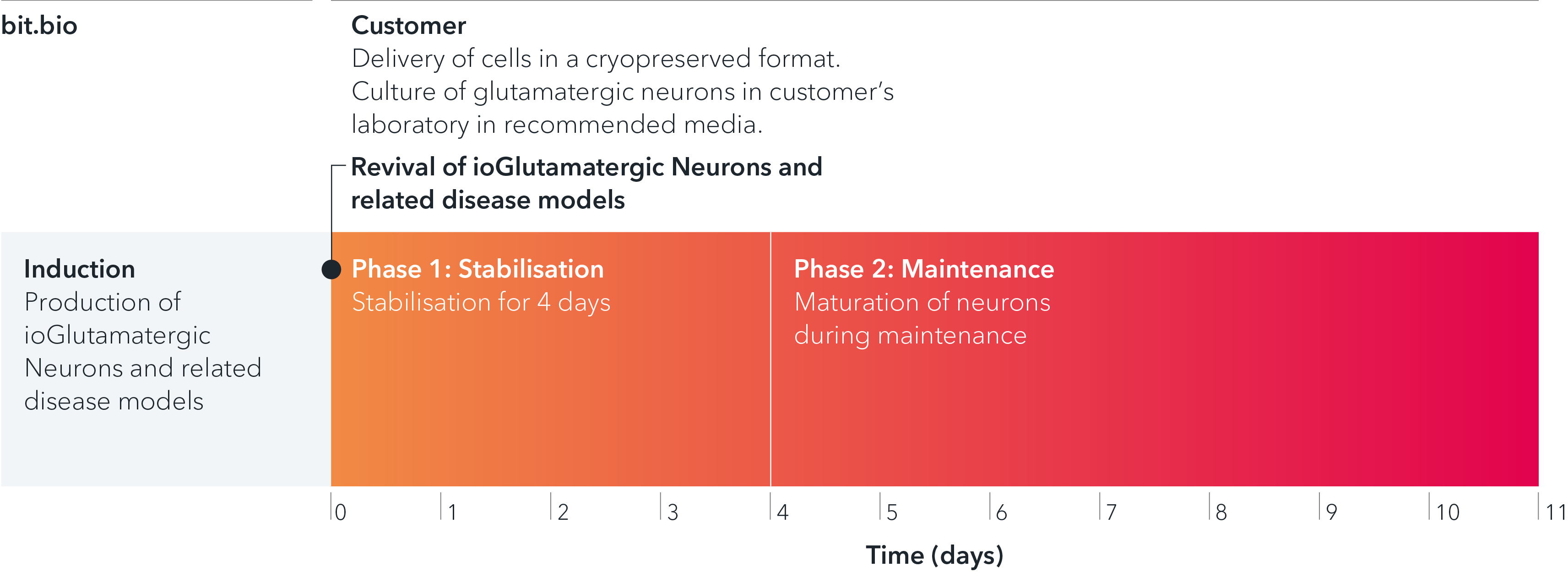
ioGlutamatergic Neurons PRKN R275W/WT are delivered in a cryopreserved format and are programmed to mature rapidly upon revival in the recommended media. The protocol for the generation of these cells is a two-phase process: Phase 1, Stabilisation for 4 days; Phase 2, Maintenance, during which the neurons mature. Phases 1 and 2 after revival of cells are carried out by the customer.
Industry leading seeding density
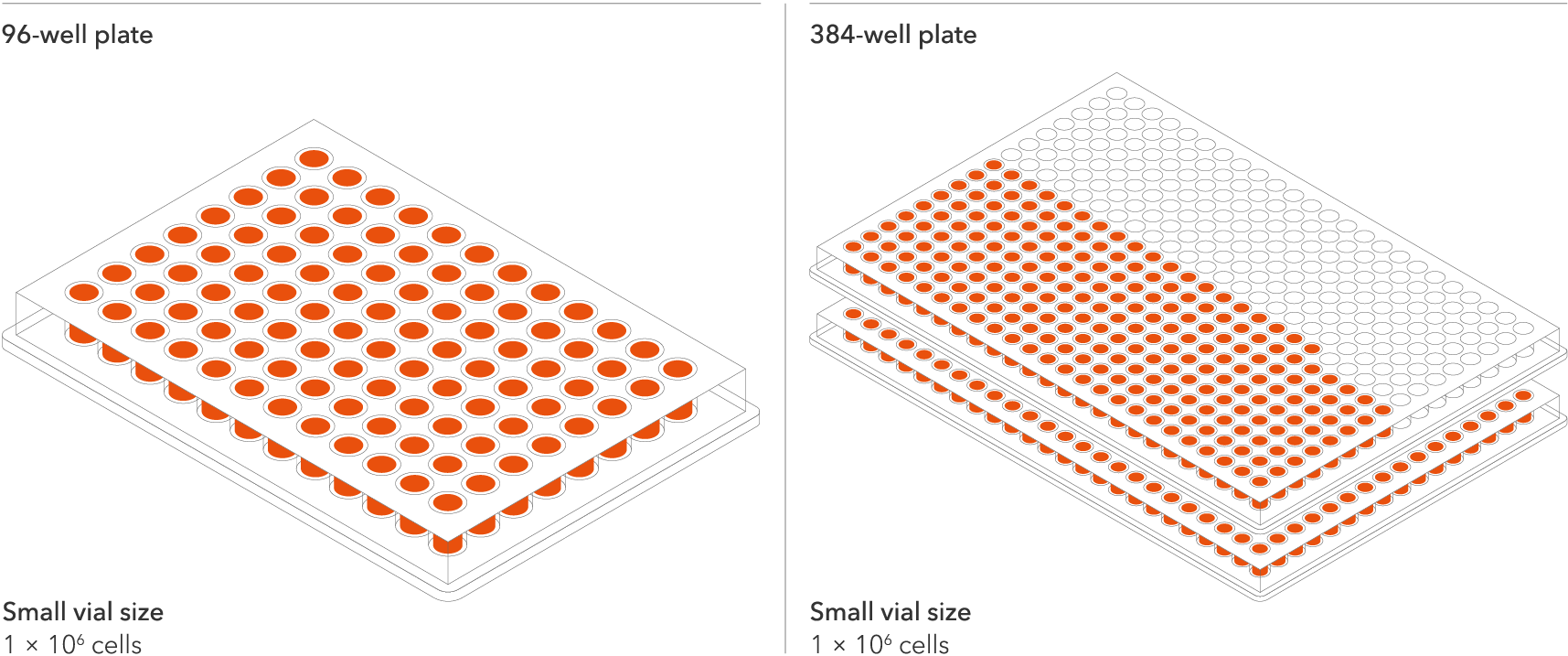
The recommended minimum seeding density is 30,000 cells/cm2, compared to up to 250,000 cells/cm2 for other similar products on the market. One small vial can plate a minimum of 0.7 x 24-well plate, 1 x 96-well plate, or 1.5 x 384-well plates. This means every vial goes further, enabling more experimental conditions and more repeats, resulting in more confidence in the data.
Starting material
Human iPSC line
Karyotype
Normal (46, XY)
Seeding compatibility
6, 12, 24, 48, 96 & 384 well plates
Shipping info
Dry ice
Donor
Caucasian adult male (skin fibroblast)
Vial size
Small: >1 x 106 viable cells
Quality control
Sterility, protein expression (ICC), gene expression (RT-qPCR) and genotype validation (Sanger sequencing)
Differentiation method
opti-ox deterministic cell programming
Recommended seeding density
30,000 cells/cm2
User storage
LN2 or -150°C
Format
Cryopreserved cells
Genetic modification
Heterozygous R275W missense mutation in the PRKN gene
Applications
Parkinson's disease research
Drug discovery and development
Disease modelling
Product use
ioCells are for research use only
Supporting documentation
Product resources
Inês Ferreira | Senior Product Manager | bit.bio
bit.bio
V11
bit.bio
2024
bit.bio
2024
Professor Deepak Srivastava
Professor of Molecular Neuroscience and Group Leader, MRC Centre for Developmental Disorders
King’s College London
Emmanouil Metzakopian | Vice President, Research and Development | bit.bio
Javier Conde-Vancells | Director Product Management | bit.bio
Dr Ania Wilczynska | Head of Computational Genomics | Non-Clinical | bit.bio
Cell culture hacks | human iPSC-derived glutamatergic neurons
Read this blog on glutamatergic neuron cell culture for our top tips on careful handling, cell plating and media changes to achieve success from the outset.
.png?width=1860&height=1260&name=bit.bio_3x2_ioGlutamatergic%20Neurons_MAP2_Hoescht_x20_hi.res%20(1).png)
Wild Type and Isogenic Disease Model cells: A true comparison.
Further your disease research by pairing our wild type cells with isogenic disease models.




Hoescht(blue)TUBB3(blue)_day4.jpg?width=604&name=bit.bio_ioGlutamatergic%20Neurons_60xMAP2(red)Hoescht(blue)TUBB3(blue)_day4.jpg)